Antagonistic Pairs
What you need to know (based on the AQA specification)
What you need to know (based on the AQA specification)
Muscles act in antagonistic pairs against an incompressible skeleton
The first key concept to understand is that muscles work in antagonistic pairs. The word antagonist means, ‘a person that opposes the other’, and muscle pairs work in this way - they oppose each other.
We are going to look at a bicep curl to see how this works. Here, the group of antagonistic muscle pairs includes the biceps & triceps. Other structures to be aware of:
Tendons
Connects muscles to bones, allowing movement of the skeleton (e.g. lifting the arm). You may have heard of tendonitis, a common sports injury (e.g. achilles tendonitis). The suffix ‘itis’ means inflammation, so tendonitis means inflammation of a tendon.
Ligaments
Connects bone to bone, providing stability at joints. A common sports injury is tearing the anterior cruciate ligament (ACL) in the knee.
Bicep Curl Example
- Pulling the lower arm upwards:
- When the bicep contracts (this is known as the agonist), the tricep does the opposite: it relaxes (this is called the antagonist) and the lower arm is pulled upwards.
- Pulling the lower arm downwards:
- When the tricep contracts (this time it’s the agonist), the bicep relaxes (the antagonist) and the lower arm is pulled downwards.
Does this work for other muscle pairs? Absolutely! Lots of examples, e.g the quadriceps and hamstrings, can pull the leg upwards or downwards.
Important
Due to how muscles are structured they pull, they do NOT PUSH
Structure of Muscles
What you need to know (based on the AQA specification)
What you need to know (based on the AQA specification)
Gross and microscopic structure of skeletal muscle. The ultrastructure of a myofibril.
Let’s continue with the bicep curl as an example, and try to understand what is happening at a structural level that is causing the muscle to contract.
High Level Organisation
- Skeletal muscle is a highly organized tissue.
- Muscles are made of bundles, called fascicles (which are encased and separated by layers of connective tissue)
- These fascicles are made of muscle fibres (again muscle fibres are separated from each other by connective tissue)
- These muscle fibres are made of myofibrils
A single muscle contains hundreds to thousands of myofibrils. Interestingly, if you are more trained (i.e you have done more bicep curls) you can have more myofibrils than someone who hasn’t hit the gym in a while.
Myofibrils are made of sarcomeres which are repeating units and the basis of muscle contraction i.e when these sarcomeres contract, this causes the entire muscle to contract.
| Muscle Fascicle -> Muscle Fibre | Myofibril |
|---|---|
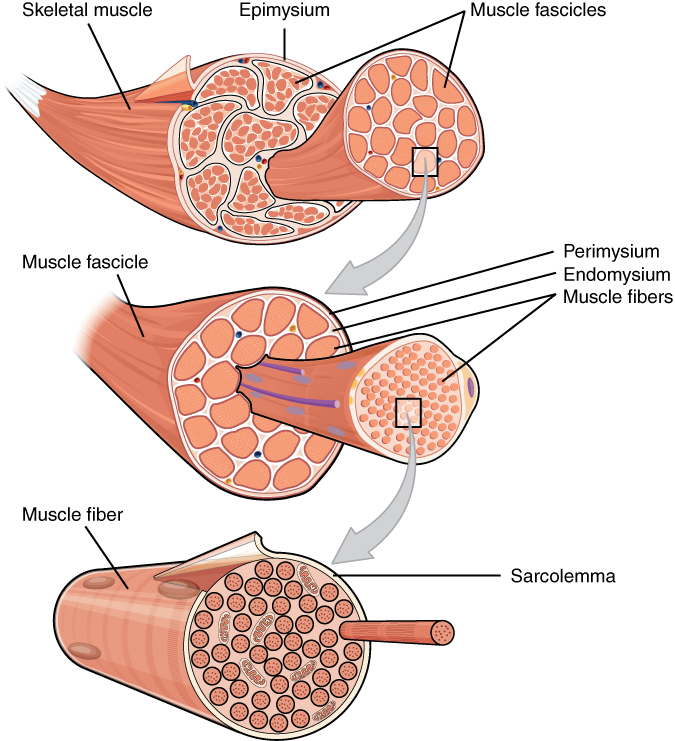 |  |
Images adapted from OpenStax College, via Wikimedia Commons (CC BY 4.0).
Muscle Fibre Structures
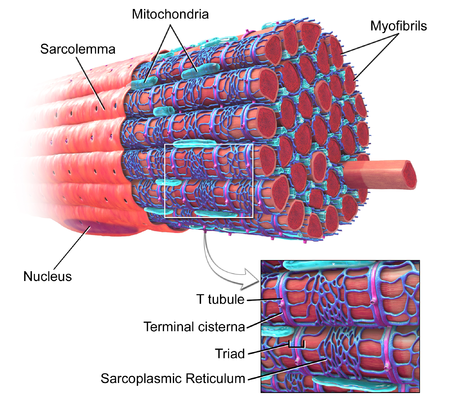
Sarcolemma
The membrane surrounding the entire muscle fibre (not individual myofibrils).
Sarcoplasm
Cytoplasm of muscle cells, which contains other structures (e.g. sarcoplasmic reticulum, nuclei)
Nuclei
The muscle fibre contains many nuclei, allowing high rates of protein synthesis for actin and myosin.
Mitochondria
Required for ATP production, which is especially important for muscle contraction.
T-tubules
Deep infoldings of the sarcolemma that transmit action potentials to the sarcoplasmic reticulum.
Sarcoplasmic reticulum
Controls calcium release; calcium triggers muscle contraction.
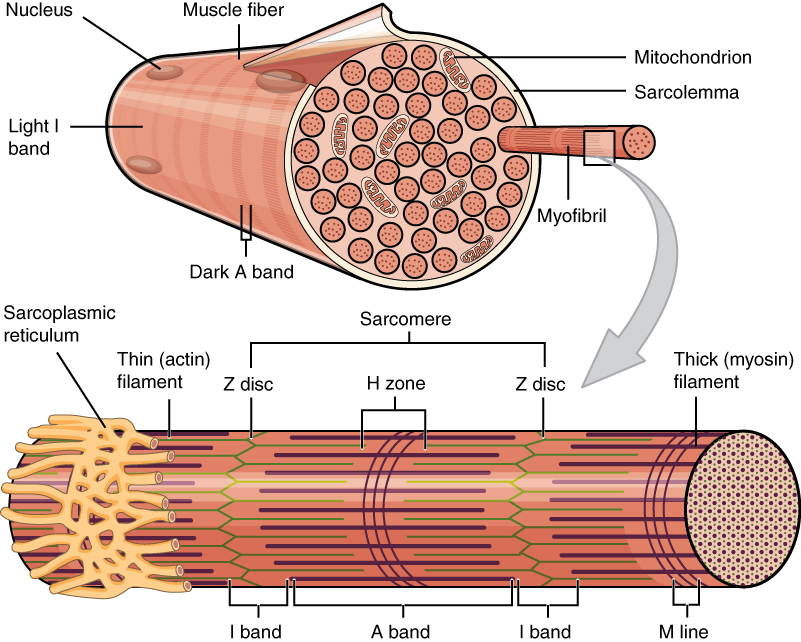
Ultrastructure of Myofibril
Our understanding of muscles came from observing myofibrils under a microscope.
Scientists observed 2 main protein filaments actin (thin filament) and myosin (thick filament).
The animation below shows you an example of 1 sarcomere unit, but in reality there are many repeating sarcomeres. You need to be aware of the following lines/bands - these can be quite challenging to remember!
M lines
Denote the middle (M for middle) of the sarcomere and provide attachment points for myosin filaments.
Z lines
Outline the boundaries of the sarcomere.
I bands
Contain actin only. Because there is a single filament present, this region appears lighter under a microscope.
A band
Region where actin and myosin overlap. This area appears darker under a microscope due to the presence of two filaments.
H zone
Contains myosin only. This region appears lighter under a microscope because only one filament is present.
What happens to these bands when the muscle contracts?
What happens to these bands when the muscle contracts?
- Z lines moved closer together (this shows that the sarcomere has shortened).
- I bands (actin only) have shortened
- H zone (myosin only) has shortened
- A band (actin & myosin) remains unchanged.
Important
- Actin and myosin filaments are sliding over each other when the muscle contracts - actin and myosin are not shortening. Only the sarcomere has shortened.
- Regions which appear darker under a microscope have 2 filaments (e.g. A band), regions which appear lighter have only 1 filament (e.g. I band/H zone)
Neuromuscular Junction
What stimulates the muscles to contract?
Let’s look at the neuromuscular junction to show how the release of calcium controls this and how muscle contraction is brought about.
Note: There is a separate topic which goes into more detail on synapses, I’m outlining it here as it relates to muscle contraction.
Steps at neuromuscular junction to cause muscle contraction
First, let’s imagine you want your bicep to contract. I’m simplifying this, but a signal is sent from your brain via neurons until it reaches this neuromuscular junction.
- This signal arrives as an action potential arrives at axon terminal (end of) the motor neurone.
- This causes opening of voltage-gated calcium ion channels which releases calcium into pre synaptic cell.
- Acetylcholine vesicles then bind to the pre synaptic membrane.
- Release acetylcholine into the synaptic cleft via exocytosis.
- Acetylcholine diffuses across the synaptic cleft.
- Acetylcholine binds to acetylcholine receptors on sarcolemma.
- This causes Na+ influx & depolarisation of sarcolemma.
- This depolarisation (if the threshold is reached) causes an action potential which travels down through t-tubules.
- This causes the sarcoplasmic reticulum to release calcium ions.
- These calcium ions cause muscle contraction - in the animation you can see the sarcomere shortening
Sliding Filament Theory
What you need to know (based on the AQA specification)
What you need to know (based on the AQA specification)
The roles of actin, myosin, calcium ions and ATP in myofibril contraction. The roles of calcium ions and tropomyosin in the cycle of actinomyosin bridge formation. (The role of troponin is not required.)
Now let’s look in more detail as to how this sarcomere contracts, specifically the sliding filament mechanism.
Who are the main players involved?
- Actin (thin filament): Two intertwined strands of actin molecules; often looks like two strands of beads.
- Myosin (thick filament): Made of a globular head + long helical tail; the head interacts with actin.
- Tropomyosin: Regulatory protein around actin; controls muscle contraction.
- Troponin: Regulatory protein; not required for AQA, but useful for understanding this concept.
Steps in Sliding Filament Theory
- Actin-myosin binding sites are blocked: When muscle is resting the tropomyosin has blocked actin-myosin binding sites, which means that myosin can’t bind to actin and muscle contraction can’t happen.
- But, when the calcium is released from sarcoplasmic reticulum (what we just looked at in neuromuscular junctions) it binds to troponin. This causes the tropomyosin to change positions exposing the actin-myosin binding sites.
- The myosin heads can now bind to actin, and a cross bridge is formed.
- Next, the myosin heads perform a power stroke which pulls the actin filaments towards the centre of the sarcomere, releasing ADP & Pi (you can see on the animation that the sarcomere has shortened)
- Next, ATP binds to the myosin head and causes it to detach from actin.
- ATP hydrolysis then occurs releasing energy to re-cock the myosin head back to original position, so that the cycle can continue.
- What happens if there is no ATP?
- What happens if there is no ATP?
Rigor mortis is a condition where there isn’t any ATP, so the myosin heads remain attached to the actin, locking the muscle fibres in a contracted position
Does this cycle keep repeating itself? When does it stop?
Does this cycle keep repeating itself? When does it stop?
- This cycle can continue assuming the muscle fibre is continuously stimulated by nerve impulses.
- As this will trigger the ongoing release of calcium ions from the sarcoplasmic reticulum (think back to the neuromuscular junction)
- The cycle stops when the muscle fibre is not stimulated and calcium returns back to sarcoplasmic reticulum via active transport, which requires ATP
Rowing Boat Analogy
- Sometimes the Sliding Filament Theory can be hard to visualise, so a rowing boat analogy is often used to help students remember
- Myosin heads are the oars of the boat
- Actin filaments are the water
- The power stroke is the pulling motion. When the myosin head changes position, it pulls the actin past the myosin
Role of ATP & Phosphocreatine
What you need to know (based on the AQA specification)
What you need to know (based on the AQA specification)
The roles of ATP and phosphocreatine in muscle contraction
Role of ATP in Muscle Contraction
-
The hydrolysis of ATP provides energy to re-cock the myosin head back into the high-energy position, which then drives the power stroke — pulling the actin filaments towards the centre of the sarcomere
-
ATP is needed to detach the actinomyosin bridge and re-set the myosin head back to its starting position.
-
Finally, it’s responsible for active transport of calcium ions to return to sarcoplasmic reticulum, when the muscle is relaxed
(Note: ‘Actinomyosin bridge’ is the terminology direct from the AQA specification, it means the link between myosin and actin. Often referred to as ‘cross-bridge’)
What are the sources of ATP?
We’ve just seen how important ATP is for muscle contraction, but how is it made?
There are 3 main sources of ATP:
- Aerobic respiration
- Anaerobic respiration (glycolysis)
- Phosphocreatine
Aerobic respiration requires oxygen and takes a lot longer to produce ATP, making it unsuitable for quick short bursts of high intensity exercise (e.g. sprinting from a lion, because that is a regular occurrence!).
High intensity exercise is fuelled partially by glycolysis and by phosphocreatine which is stored in muscles.
| Aerobic Respiration | Anaerobic Respiration | Phosphocreatine |
|---|---|---|
| Oxygen required | No oxygen needed Lactate is produced | No oxygen needed Phosphocreatine + ADP ⇌ Creatine + ATP |
| Produces a lot of ATP (~32 ATP) | Produces much less ATP than aerobic (~2ATP) | (~1ATP) |
| Produces ATP at slower rate | Produces ATP at a fast rate | Regenerates ATP very rapidly |
| Low intensity exercise | High intensity exercise (1-3 mins) | Very high intensity short burst < 10 seconds |
Phosphocreatine
- Is a phosphorylated form of creatine, stored in the muscles.
- It is used to rapidly regenerate ATP for high intensity exercise (e.g short sprints, 100m)
Phosphocreatine
Phosphocreatine (PCr) donates a phosphate group to ADP to form ATP and Creatine
Phosphocreatine + ADP ⇌ Creatine + ATP
- It’s a reversible reaction, and the stores of phosphocreatine become replenished during rest.
- Interestingly
- Some people take creatine supplements to help increase stores of phosphocreatine, to improve their performance at very high intensity exercise
- If you wanted to train your phosphocreatine system (i.e high intensity/short sprints) it’s important to take long enough rests in between sprints to ensure you can replenish your phosphocreatine stores
Slow & Fast Twitch Muscle Fibres
What you need to know (based on the AQA specification)
What you need to know (based on the AQA specification)
The structure, location and general properties of slow and fast skeletal muscle fibres.
There are two main types of muscle fibres: Slow & Fast Twitch.
These exist in different proportions across individuals and across different types of muscles.
For example, sprinters often have a higher proportion of fast twitch muscle fibres than ultramarathon runners. The relative proportions also vary depending on the location of the muscle in the body (e.g. calf muscles often have a higher proportion of slow twitch muscles as they need to be able to withstand continuous muscle contraction).
Slow Twitch
Function
- Contract more slowly than fast twitch
- Less powerful contractions over a long period of time
- Used for longer sustained endurance activities
Structural adaptations
- More myoglobin (protein that acts as an oxygen store, appears bright red)
- More mitochondria for aerobic respiration to produce more ATP
- Rich supply of blood vessels to deliver oxygen and glucose for aerobic respiration
- High resistance to fatigue (relies on aerobic respiration, generates large amounts of ATP)
Fast Twitch
Function
- Contract quickly
- More powerful contractions over a short period of time
- Used for shorter, higher intensity activities
Structural adaptations
- Increase in size (hypertrophy) so they are able to generate powerful contractions
- High stores of phosphocreatine
- High stores of glycogen
- Fatigues quickly (relies on anaerobic respiration, lactate is produced)
Structure & Function linkage
Try to think about how the structure can enable the function. E.g Fast twitch fibres need to contract quickly and forcefully, so a structure that could support that would be larger (hypertrophy of muscles)
Exam Question Practice
Which statement correctly describes the changes in length in a sarcomere when a myofibril contracts?
Fast muscle fibres have a higher concentration of glycogen than slow muscle fibres.
Explain how the difference in glycogen concentration is related to the different properties of these muscle fibres.
(4 marks)Hint
Think about what type of respiration each fibre type uses and how glycogen relates to ATP production.
Mark Scheme
- Fast fibres contract quickly (used for short, high-intensity bursts), while slow fibres contract slowly (used for sustained, lower-intensity activity) (1 mark)
- Fast fibres rely mainly on anaerobic respiration (using glycogen as fuel), while slow fibres rely on aerobic respiration (1 mark)
- Anaerobic respiration produces ATP rapidly but yields less ATP per glucose molecule — so fast fibres need large glycogen stores to sustain bursts of activity (1 mark)
- Glycogen is hydrolysed to glucose (glycogenolysis) — this glucose is the substrate for respiration in fast fibres (1 mark)
Tips from examiner reports
- You needed to provide a comparison between fast fibres and slow fibres in terms of their speed of contraction
- You need to refer to glycogen as a store of glucose or to the hydrolysis of glycogen to glucose
- Don’t say ‘energy produced’, this wasn’t credited
ATP is essential for muscle contraction.
Describe the roles of ATP in muscle contraction.
(2 marks)Hint
Think about the different stages where ATP is used in the sliding filament mechanism.
Mark Scheme
- ATP is needed to detach the myosin head from actin — breaking the actinomyosin bridge (1 mark)
- ATP hydrolysis provides energy to re-cock (reset) the myosin head to its high-energy position, so it can perform the next power stroke and pull actin filaments inwards (1 mark)
- ATP is also needed for the active transport of calcium ions back into the sarcoplasmic reticulum when the muscle relaxes (1 mark)
Describe and explain how taking creatine supplements and ‘carbohydrate loading’ can improve performance of different types of muscle fibres during different types of exercise.
(5 marks)Hint
Consider which type of muscle fibre benefits from each supplement and why.
Mark Scheme
- Fast muscle fibres are used during short-term, high-intensity exercise (e.g. sprinting) (1 mark)
- Slow muscle fibres are used during longer-term, lower-intensity exercise (e.g. marathon running) (1 mark)
- Creatine supplements increase the amount of phosphocreatine stored in fast muscle fibres (1 mark)
- Phosphocreatine donates a phosphate group to ADP: phosphocreatine + ADP → creatine + ATP — rapidly regenerating ATP for high-intensity bursts (1 mark)
- Carbohydrate loading increases glycogen stores in muscle — glucose is converted to glycogen (glycogenesis) for storage (1 mark)
- During exercise, glycogen is hydrolysed back to glucose (glycogenolysis) to provide a fuel source (1 mark)
- This glucose is used in respiration to produce ATP for slow or fast muscle fibres (1 mark)
Tips from examiner reports
- Students frequently associated the type of muscle fibre with aerobic or anaerobic exercise which was not part of the required explanation
- Abbreviations for phosphocreatine are not in the specification and should not be used in examinations (you can only use abbreviations which are on the specification)
- Better responses included the storage of glucose as glycogen (Not starch!)
Figure 3 shows the banding pattern of a single sarcomere.
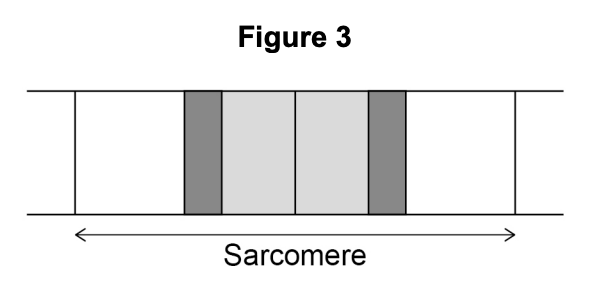
Explain the banding pattern shown in Figure 3.
(3 marks)Hint
Think about which filaments (actin and/or myosin) are present in each region and how that affects appearance.
Mark Scheme
- The light I band contains actin filaments only — one type of filament makes this region appear lighter (1 mark)
- The H zone contains myosin filaments only — also just one type of filament, so it also appears lighter (1 mark)
- The darkest region is where actin and myosin overlap — two types of filament in the same zone create a darker appearance (1 mark)